  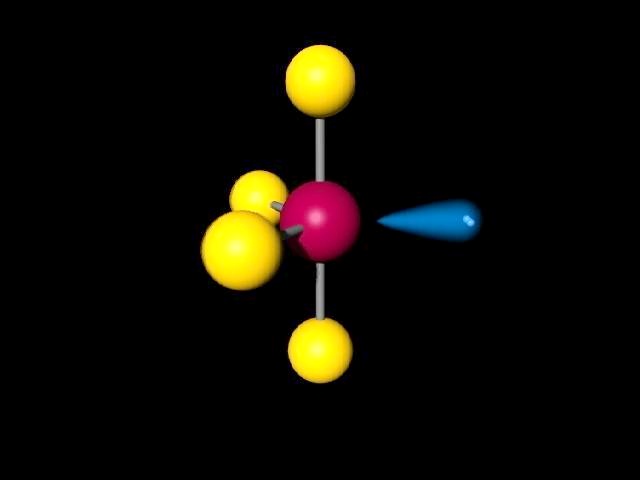
Repulsion by bonding pairs at 120° is much smaller and less important. An equatorial lone pair is repelled by only two bonding pairs at 90°, whereas a hypothetical axial lone pair would be repelled by three bonding pairs at 90° which would make it stable. Some common shapes of simple molecules include: Linear: In a linear model, atoms are connected in a straight line. This is true because the lone pair occupies more space near the central atom (A) than does a bonding pair of electrons. A bond angle is the geometric angle between two adjacent bonds. H30+ Molecular geometry Valence electrons 5. CIF, Molecular Geometry: Bond Angle (s): Lewis Structure: El. With one lone pair of valence electrons, you get a seesaw molecular geometry. Using VSEPR, determine the electron pair geometry, the molecular geometry, and the bond angle (in degrees) for each compound. 120 for equatorial bonds and 90 for axial bonds. As a result, you have 5 electron groups, so the electron geometry would be trigonal bipyramidal. Trigonal pyramidal molecules, while still adhering to the VSEPR, have bond angles that are bent out of their expected angles, and they can vary (<109 degrees). Solution for QUESTION 1 A molecule with a seesaw molecular geometry has a bond angle of <90. An atom bonded to 5 other atoms (and no lone pairs) forms a trigonal bipyramid with two axial and three equatorial positions, but in the seesaw geometry one of the atoms is replaced by a lone pair of electrons, which is always in an equatorial position. The site preference for larger domains to go equatorial also affects the bond lengths and bond angles in molecules. Therefore, you can put 6x4 on each fluorine, 2x4 to account for four single bonds, and 2 for the last 2 valence electrons available. The seesaw geometry occurs when a molecule has a steric number of 5, with the central atom being bonded to 4 other atoms and 1 lone pair (AX 4E 1 in AXE notation). As for 3bp 2lp, the geometry can be trigonal planar (in which case the bond angles are 120) or T-shape - in which case the bond angle is approximately 85 as. Quick facts: Seesaw molecular geometry, Examples, Point gr. Seesaw Molecular Geometry/Shape and Bond Angles Seesaw molecular geometry - Wikipedia Seesaw Molecular Geometry - Seesaw Shaped Molecules, Chapter 9 PLQs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
